|
8.
Evaluation of the Activated Carbon Prepared from
Algae Gracilaria for the Biosorption of Cu(II) from Aqueous
Solutions
 Akbar
Esmaeili*, Akbar
Esmaeili*, Samira
Ghasemi, Samira
Ghasemi,
 Abdolhossein
Rustaiyan Abdolhossein
Rustaiyan
 Department
of Chemical Engineering, North Tehran Branch, Islamic Azad
University, Tehran, Iran. Department
of Chemical Engineering, North Tehran Branch, Islamic Azad
University, Tehran, Iran.
 Department
of Marin Science and Technology, Islamic Azad University, North
Tehran Branch, Tehran, Iran. Department
of Marin Science and Technology, Islamic Azad University, North
Tehran Branch, Tehran, Iran.
Corresponding author:
akbaresmaeili@yahoo.com Tel : +989121484813
ABSTRACT
In this study, the batch
removal of copper(II) ions from aqueous solution and wastewater
using marine red algae Gracilaria was investigated. Activated carbon
prepared from Gracilaria by acid decomposition was used for this
purpose. Activated carbon prepared marine alga were used as a low
cost sorbent. The effect of pH, biosorption time, adsorbent dose,
and metal ions concentration, were studied. The most effective pH
was found to be 4.0. At the optimum pH (pH=4), the biosorption
capacities were found in the range of 95.53 %, 93.72 % and 88.84 %,
depending on initial concentration of 70, 100 and 150 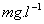 ,
respectively. The optimum time was found to be 2 hours. The
equilibrium adsorption data are fitted to Langmuir and Freundlich
isotherm models. Both the models represent satisfactorily the
experimental data. The adsorption follows second order kinetic. This
study shows that activated carbon prepared from algae is a valuable
material for the removal of copper from aqueous solution and
wastewater and a better substitute of using in activated marine red
algae. ,
respectively. The optimum time was found to be 2 hours. The
equilibrium adsorption data are fitted to Langmuir and Freundlich
isotherm models. Both the models represent satisfactorily the
experimental data. The adsorption follows second order kinetic. This
study shows that activated carbon prepared from algae is a valuable
material for the removal of copper from aqueous solution and
wastewater and a better substitute of using in activated marine red
algae.
Keywords: Biosorption,
copper, Gracilaria, wastewater, activated carbon, isotherm
model.
<<< |

